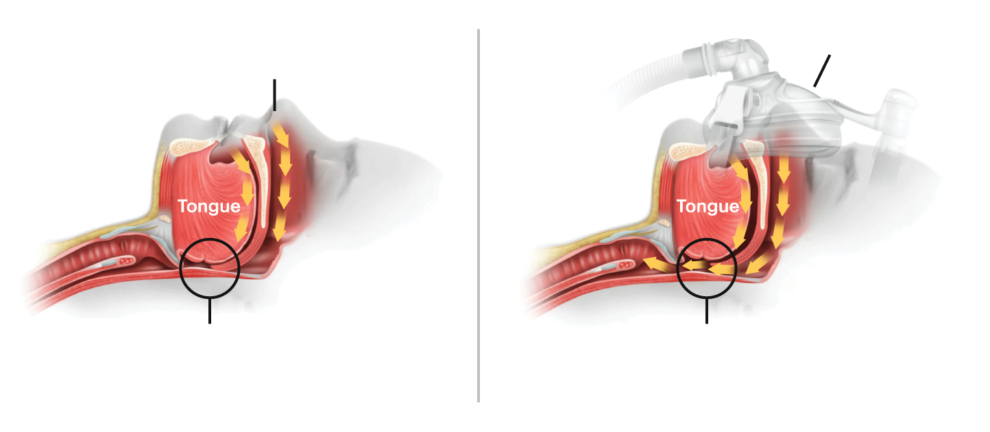
Philips failed to report at least 3,700 complaints about its sleep apnea devices and respirators to the United States regulator FDA since 2010, NRC, research collective ProPublica, and the Pittsburgh Post Gazette reported based on their analysis of over 100,000 public reports.
According to the researchers, the first complaint came in 2010, shortly after Philips started using a sound-dampening foam in the breathing aids that triggered a mass recall in 2021. At the recall, Philips said the sound-dampening foam could disintegrate when it made contact with certain cleaning agents, resulting in users potentially breathing in carcinogenic substances.
United States law dictates that device makers must inform the government within 30 days if they receive reports of patient injuries, deaths, or malfunctions that could cause harm. In the 11 years between the launch of the sound-dampening foam and the recall, Philips received at least 3,700 such reports in the United States that it kept in its files and didn’t report to the FDA, according to the researchers.
The withheld complaints contain at least 10 reports about patients who may have died due to the use of a ventilator. Other reports described “black particles” or “dirt and dust” inside the machines. One described an “oily-like substance,” another spoke of “black shavings in the chamber,” and another said it was “contaminated with unknown sticky substance.”
U.S. law also states that device makers must immediately investigate reported faults that may cause harm. Philips launched its first investigation in 2019. In 2021, it recalled 15 million DreamStation sleep apnea machines and ventilators that used the foam in question. The FDA confirmed to the researchers that Philips had withheld many reports. According to the American regulator, Philips found “a large number of foam disintegration complaints that should have been sent to the FDA” during “retrospective reviews” following the recall.
A Philips spokesperson told NRC that “out of an abundance of caution,” the company sent old complaints that “may be related to the deteriorated foam” to the FDA despite “previously determining that these complaints did not need to be reported.” According to Philips, it was unaware of the scope of the problem because, until early 2021, the complaints were handled “one by one by Philips Respironics” – the Philips subsidiary in Pittsburg.
Because Philips held the complaints under wraps for over a decade, the regulator only intervened eleven years after the company got its first report. Through the spring of 2021, the FDA had only received 30 reports from the Pittsburg subsidiary of incidents with disintegrating foam.
The recall caused Philips serious financial troubles. Its stock price has halved, CEO Frans van Houten resigned, and his successor Roy Jakobs has announced two major rounds of layoffs. Lawyers in the United States are preparing mass claims amounting to billions of euros against the company.
Earlier this month, Philips announced that it had reached a settlement in the U.S. to resolve over 500 pending lawsuits and damage claims due to the recall. In April, Philips announced that it set aside 575 million euros to cover the potential cost resulting from lawsuits and settlements in the country.
Source: NL TIMES